The rapid evolution of silicon-carbon (Si-C) composite anodes and sodium-ion batteries has placed a spotlight on the importance of microporous carbons. In Si-C anodes, high microporosity provides a robust scaffold for silicon deposition, maximizing specific surface area and capacity. For sodium-ion batteries, hard carbon remains the leading candidate for efficient ion intercalation.
A wide range of carbon precursors can be used to produce porous carbon-based anodes, including coal-derived carbon, pitch-derived carbon, polysaccharide-derived carbon, and biomass-derived carbon. Among these, utilizing biomass waste, such as fruit shells, peels, straw, and rice husk, through high-temperature carbonization and structural restructuring to prepare high-quality microporous anode materials is an economical and environmentally friendly approach that aligns with global resource recycling and utilization policy.
Advanced Micropore Characterization via Dual-Gas Adsorption
The use of microporous carbon anode materials necessitates reliable characterization of micropores using advanced gas adsorption techniques. Standard N, gas adsorption instruments often fall short when characterizing the finest micropores due to the absence of low-pressure transducers. To address this, Micromeritics utilizes a dual-probe approach on its industry-leading TriStar II Plus 3030 instruments:
- Nitrogen (N2) Adsorption: Provides a broad view of mesopores and larger micropores.
- Carbon Dioxide (CO2) Adsorption (at 273 K): CO2 is a more linear and smaller molecule (3.3 Å) compared to N2 (3.64 Å), enabling its diffusion into smaller pores. At 273K, due to the faster kinetics and the high saturation vapor pressure, CO2 diffuses more rapidly into narrow carbon pores, probing smaller pore sizes more efficiently
The use of CO₂ as a probe molecule, together with the advanced NLDFT (Non Local Density Functional Theory) model, enables precise determination of pore size distributions across a broad pore range.
Case Study: Investigation of biomass-derived hard carbon anode materials
N2 Adsorption isotherm of a biomass- derived hard carbon anode material using a Tristar II Plus 3030 is shown in Figure 1 below.
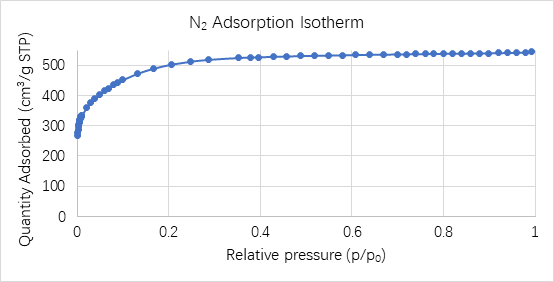
Figure 1: N2 adsorption isotherm of biomass-derived microporous hard carbon anode
This is a typical type Ib adsorption isotherm, indicating the presence of micropores and 2-3 nm mesopores in this material. The overall adsorption capacity is also relatively large, and the material has a high pore surface area.
During Bulk testing (Figure 2), an even more differentiating result was generated by the Permeability test. Metal Powder C generates a significantly higher Pressure Drop across the powder bed than the other samples, indicating that Metal Powder C is considerably less permeable than Powders A and B.
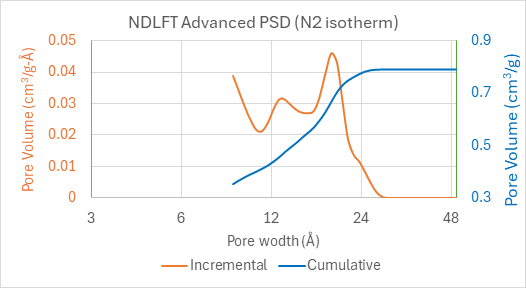
Figure 2: pore size distribution calculated from N2 isotherm in biomass-derived microporous hard carbon anode
The pore size distribution calculated using the NDLFT analysis of the N2 isotherm is shown in Figure 2. It reveals pores ranging from 9 Å to 28 Å. Pores below 9 Å are not revealed from this N2 isotherm. On a TriStar instrument, microporosity can be investigated by collecting an isotherm with CO2 in addition to the N2 isotherm.
Tristar allows easy switching up to 3 gases, in addition to a dedicated port for Helium. The CO2 adsorption isotherm measured at 273 K is shown in Figure 3.
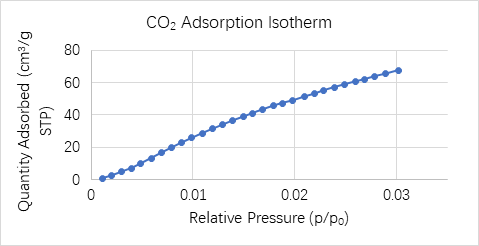
Figure 3: CO2 adsorption isotherm of biomass -derived microporous hard carbon anode
It is well reported in literature that a dual gas NLDFT model can provide a comprehensive analysis of pores in microporous materials1-3. By applying the dual gas NLDFT Advanced PSD method in the MicroActive software – HS-2D-NLDFT for N2 and NLDFT for CO2 – we can obtain the complete pore-size distribution covering the micro and mesopore ranges, as shown in Figure 4.
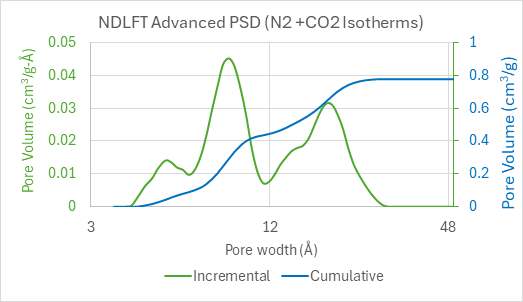
Figure 4: Combined pore size distribution of N2 and CO2 in biomass-derived microporous hard carbon anode
In this figure, the green curve represents the pore size distribution, with the size corresponding to the peak being called the modal pore size. The blue curve represents the cumulative pore volume. This analysis shows that this material is mainly composed of micropores smaller than 20 Å, and some small mesopores in the 20-30 Å range. This analysis also reveals that the cumulative specific surface area in the pores of this material reaches 1525 m²/g. In microporous materials, the surface area calculated with NLDFT is known to be more accurate than a simple BET estimation.

Total pore volume and pore surface area of biomass-derived microporous hard carbon anode
Anode material with such a large surface area is a good template to prepare high-silicon-content silicon-carbon anodes, thereby greatly improving the anode‘s specific capacity.
Model Accuracy and Agreement
As shown in Figure 5, the NLDFT Advanced PSD model shows excellent agreement between the experimental data points and the theoretical adsorption isotherms (solid lines). This validation confirms the reliability of the derived pore volumes and surface area measurements.
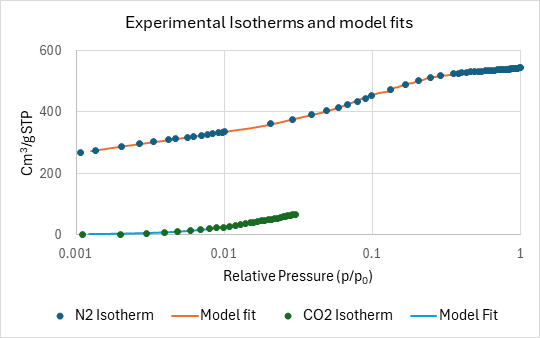
Figure 5: Degree of agreement between experimental isotherms and theoretical adsorption isotherms
Conclusion
The biomass-derived hard carbon used in this case study demonstrates exceptionally high surface area, making it an ideal precursor for high-silicon-content anodes. By converting waste into high-value energy storage materials, manufacturers can significantly reduce costs while enhancing specific capacity.
Micromeritics continues to lead in battery material characterization, providing the advanced hardware and analytical models, like the TriStar II Plus 3030 instruments with multiple gas options, and the HS-2D-NLDFT-based analysis model, to reliably analyse the surface area, pore volume, and pore size distribution in these complex microporous materials, unlocking their potential in high-performance energy storage applications.
References